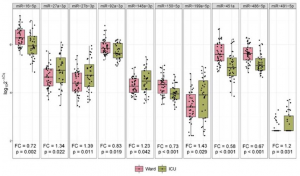
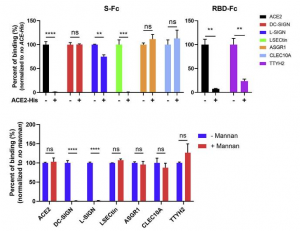
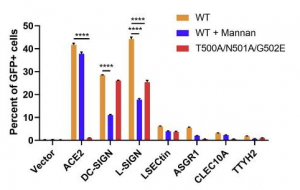
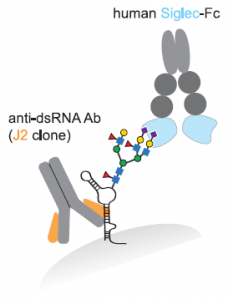
A group from National Cheng Kung University, Tainan, etc. has reported that human surfactant protein D would have therapeutic potential against SARS-CoV-2 infection.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8161545/
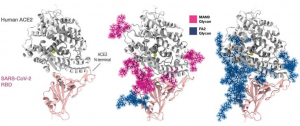
Human surfactant protein D (SP-D), a collagen-containing C-type lectin and a member of the collectin family, is known to be involved in pulmonary surfactant homeostasis and immunity. The ability of recombinant fragment of human SP-D(rfhSP-D) to inhibit infection of SARS-CoV-2 was examined using pseudotyped lentiviral particles expressing SARS-CoV-2 S1 protein.
It was found that rfhSP-D bound SARS-CoV-2 S1 protein in a dose-dependent manner; this interaction was inhibited by maltose and EDTA. No rfhSP-D binding was observed in the absence of RBD in this assay.
Inhibition of rfhSP-D binding to S protein by EDTA or maltose suggested that rfhSP-D bound to the carbohydrate moieties on S protein of SARS-CoV-2. A luciferase reporter assay with pseudotyped lentiviral particles expressing SARS-CoV-2 S1 protein was used to evaluate infectivity. Approximately 0.5 RLU fold reduction was seen with rfhSP-D (5 or 10 µg/ml) treatment comparing to untreated sample (1 RLU fold; ACE2 overexpressing HEK293T cells + SARS-CoV-2). A significantly reduced luminescent signal following rfhSP-D treatment indicated that the interaction of rfhSP-D with SARS-CoV-2-S1 restricted the binding and entry of the virus.
These results highlight the therapeutic potential of rfhSP-D in SARS-CoV-2 infection.