A group from Univ. Grenoble Alpes, France, etc. has reported a mechanism of SARS-CoV-2 infection through C-type lectin receptors (CLRs).
https://journals.plos.org/plospathogens/article/authors?id=10.1371/journal.ppat.1009576
Authors studies molecular interaction between various CLRs (DC-SIGN, L-SIGN, MGL, and Langerin) with SARS-CoV-2 spike protein using SPR. DC-SIGN, L-SIGN, and MGL showed affinities in the μMrange in terms of Kd, from around 2 to 10 μM, and that of Langerin was speculated to be weaker at least one order of magnitude.
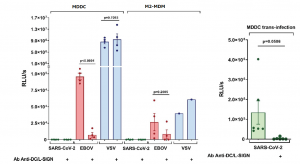
In order to understand the roles of CLRs in SARS-CoV-2 infection, infection experiments were performed using monocyte-derived dendritic cells (MDDCs), monocyte-derived macrophages (M2-MDM) and Vero E6 cells. Interestingly, VSV/SARS-CoV-2 pseudotyped virus did not infect MDDCs or M2-MDMs, despite DC-SIGN expression. In the case of VSV/EBOV-GP, however, direct infection was observed and DC-SIGN-mediated cis-infection was clearly blocked with anti-DC/L-SIGN in the case of MDDCs (92.5% inhibition of infection), followed by M2-MDM (68.4% inhibition). VSV/VSV-G also showed strong infectivity to both MDDC and M2-MDM. However, this infection was DC-SIGN independent, since anti-DC-SIGN antibodies did not impact the infection level.
To study the potential function of DC/L-SIGN in SARS-CoV-2 trans-infection, MDDCs were incubated with VSV/SARS-CoV-2 and placed onto susceptible Vero E6 cells which is the reference ACE2+ cell line for SARS-CoV-2 cell culture. Interestingly, DC-SIGN promoted efficient SARS-CoV-2 transfer from MDDC to Vero E6. An anti-DC-SIGN antibody could reduce substantially the infectivity observed (98% inhibition). These experiments confirmed that the role of DC-SIGN and L-SIGN in the process of SARS-CoV-2 could be trans-infection.