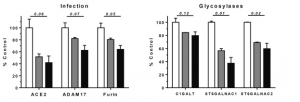
A group from Daiichi Sankyo Co., Ltd. has reported about a new chemical which is added into CHO culture media and thereby is able to improve productivity of mAbs as therapeutic drugs.
https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0250416
Authors intended to improve productivity of mAbs with controlling those glycan modification by adding a chemical into CHO culture media.
They started screening from 23,277 chemicals, and through the following 2nd screening condition; over 120% for relative mAb concentration, 105% for relative cell-specific productivity, and 80% for viability, a few candidates were selected. From the final selectins, 4-(2,5-dimethyl-1H-pyrrol-1-yl)-N-(2,5-dioxopyrrolidin-1-yl) benzamide (MPPB) was selected as a most suitable chemical to meet the intended requirement. MPPB concentrations of 0.32 to 0.64 mM were used, and with this addition, the productivity of mAbs increased by 1.5times.
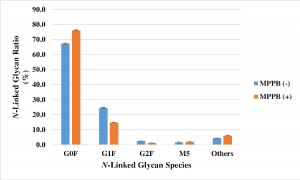
G0F was the major N-linked glycan, and G1F was decreased from 24.5 to 14.8% under the MPPB-added condition, although others were not changed