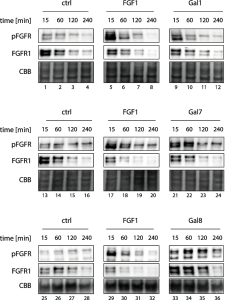
A group from Department of Biotechnology, Graduate School of Engineering, Osaka University, Osaka, Japan has reported about glycosylation of recombinant adeno-associated virus serotype 6 (rAAV6) which is used for Gene therapy.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC11107246/
Glycosylation of recombinant adeno-associated viruses (rAAVs) that are used for gene theraphy has been evaluated by Lectin microarrays.
It was found that rAAV6 was mainly O-glycosylated with the mucin-type glycans, O-GalNAc (Tn antigen), and mono- and di-sialylated Galβ1-3GalNAc (T antigen).
The purpose of this study was to see if glycosylation of rAAV6 can affect gene therapy safety in terms of immunogenecity and overall transduction efficacy of this method.
However, unfortunately, this study still could not evaluate the direct influence of mucin-type O-glycans on transduction efficiency.