A group from Genomics Research Center, Academia Sinica, Taipei, Taiwan has reported about a glycan-binding protein modulated the TGF-β-driven signaling and metastasis of colorectal cancer (CRC).
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC11375092/
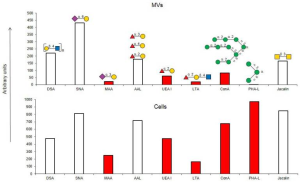
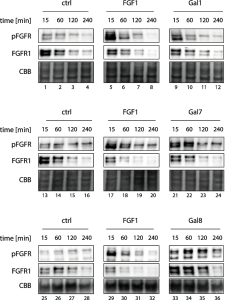
This study demonstrate that galectin-8 alters non-canonical TGF-β response in CRC cells and suppresses CRC progression, although Galectin-8 is not a canonical regand of TGF-β Receprtor.
In detail, in the absence of galectin-8, TGF-β binds to type II TGF-β receptor (TβRII), thereby promoting epithelial-mesenchymal transition (EMT).
In the presence of galectin-8, galectin-8 binds to TβRII through galactosylaeted-glycans, resulting in a decrease of TGF-β signaling-mediated EMT.
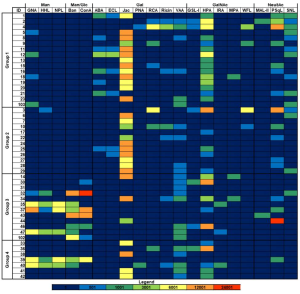
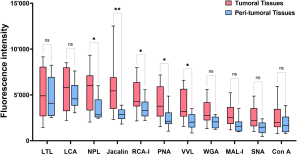
It was also shown that galectin-8 expression is downregulated during CRC progression.
The expression of galectin-8 is significantly lower in T4 stage than that in T1 stage.