A group from Laboratory of Cancer Biology and Molecular Immunology, Graduate School of Pharmaceutical Sciences, The University of Tokyo, Tokyo, Japan, etc. has reported that T-antigen (with or without sialylation) on MUC21 is required to elicit an anti-apoptotic effect.
https://pubmed.ncbi.nlm.nih.gov/35410995/
Mucins are highly O-glycosylated proteins thought to protect epithelial surfaces from physical, chemical, and biological insults, and attention to mucins has been paid in the fields of cancer diagnosis and cancer therapy. Mucin 21 (MUC21) is a unique transmembrane-type mucin, found in an endeavor to identify mouse epiglycanin, a mammary carcinoma-associated mucin.
It was first reported that expression of MUC21 by transfection makes epithelial cells resistant to apoptosis. For this effect to occur, MUC21’s O-glycans need to be extended to contain galactose (Gal) residues that may or may not be sialylated. In addition, we show that MUC21-dependent apoptosis resistance is not altered in the absence of galectin-3, suggesting that another galectin or carbohydrate-binding molecule is mediating the effect.
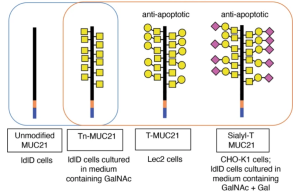
To investigate whether the extension of O-glycans with galactose is necessary for the apoptotic effect of MUC21, CHO-glycosylation variant ldlD cells were used. ldlD cells can synthesize O-glycans only after addition of GalNAc and/or galactose to the culture medium due to a deficiency in the enzyme UDP galactose and UDP-Gal/UDP-GalNAc 4-epimerase. ldlD–MUC21 transfectants grown in regular medium showed only one band at molecular weight around 160 kDa, indicating that ldlD cells expressed only nonglycosylated MUC21. When the medium was supplemented with GalNAc, bands at molecular weight around 200 kDa were seen when lysate was blotted with VVA lectin, suggesting that MUC21 carried Tn-antigen. When the medium was supplemented with GalNAc plus Gal, two bands appeared, one at approximately 200 kDa (no sialidase treatment) and one above 200 kDa (after sialidase treatment). The latter band was also seen when PNA lectin was used for blotting. These results indicate that GalNAc plus Gal supplementation led to the expression of sialyl T-antigen on MUC21.
Comparing these three kinds of samples, it was clearly shown that T-antigen (with or without sialylation) on MUC21 is required to elicit an anti-apoptotic effect.
Galectin-3 has been shown to have antiapoptotic effects in a variety of cell types. So, it was tested if galectin-3 is involved in the induction of MUC21-dependent apoptosis resistance by silencing galectin-3 in MUC21-transfectant HEK293 cells. However , it was found that galectin-3 silencing did not significantly alter the amount of apoptosis resistance in MUC21-transfectant cells.
What’s going on here?