A group from Leroy T. Canoles Jr. Cancer Research Center, Eastern Virginia Medical School, Norfolk, Virginia 23507, USA, etc. has reported about site-specific N-Linked glycopeptide characterization of Prostate-Specific Membrane Antigen from metastatic prostate cancer cells.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9435049/
The prostate-specific membrane antigen (PSMA), also known as folate hydrolase 1 (FOLH1) or glutamate carboxypeptidase 2, was previously studied as a PCa biomarker in tissues and body fluids using conventional biochemical methods with mixed results. PSMA is not exclusively expressed in prostatic tumors; the protein is expressed in low abundance in prostate epithelium, in the neo-vasculature of exclusive solid tumor types, and in some healthy tissues including proximal renal tubes, duodenum, and ganglia of the nervous system. In PCa, PSMA abundance increases with disease severity, and up to 100-fold higher abundance has been observed in advanced aggressive forms of the disease compared to normal tissue.
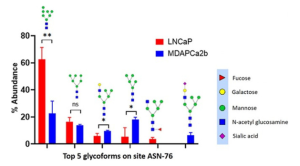
In this study, two PCa cell lines, LNCaP cells (CRL-1740) and MDAPCa2b (ATCC CRL-2422) were used to compare site-specific N-linked glycopeptide characterization of PSMA.
It was demonstrated that there are significant differences in the expression of several glycans in two cell lines LNCaP and MDAPCa2b (see below), which have different phenotypes, and further there are significantly more glycans identified in MDAPCa2b compared to LNCaP cells.
These studies will form the basis of developing site-specific PSMA glycoform-based prognostic markers for PCa disease stratification in the future.