A group from Department of Obstetrics and Gynecology, Wakayama Medical University, Wakayama 641-0012, Japan, etc. has reported on diagnostic application of C-mannosyl Tryptophan.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6924205/
C-Mannosyl tryptophan (CMW) is a glycosylated amino acid first isolated from human urine with a unique glycan structure in which an α-mannose is bound to the indole C2 carbon of a Trp residue through a C-C linkage. CMW was also identified in human ribonuclease 2 (RNase2) as a post-translational modification.
In medical field, it was first reported that blood CMW is elevated in patients with renal dysfunction, including renal diseases associated with type 2 diabetes. In cancer biology, it was recently reported that C-mannosylation of R-spondin 2 activates Wnt/β-catenin signaling and migration activity in various human tumor cells. This study suggested that C-mannosylation of R-spondin 2 is involved in the promotion of cancer progression. Furthermore, spondin 2 (mindin), a substrate protein for C-mannosylation, is increased in the blood of ovarian cancer patients. These studies suggest that protein C-mannosylation and CMW may be involved in the pathophysiological processes of cancer progression.
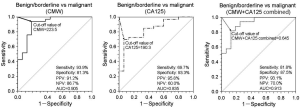
Plasma CMW in ovarian cancer was significantly higher in the malignant tumor group than in the borderline and benign tumor groups compared with healthy controls. Receiver operating characteristic curve analysis of plasma CMW distinguished malignant tumors from borderline/benign tumors (AUC=0.905). Discrimination performance was greater than that of conventional cancer antigen CA125 (AUC=0.835), and CMW + CA125 combined achieved even greater discrimination (AUC=0.913, 81.8% sensitivity, 87.5% specificity).
CMW in biological samples was analyzed and quantified by chromatographic assay. The samples were injected into an UPLC system, and CMW was quantified by measuring the fluorescence (excitation at 285 nm/emission at 350 nm).