A group of Department of Agronomy and Horticulture and Center for Plant Science Innovation, University of Nebraska – Lincoln, Lincoln, NE, USA, etc. has reported about optimal inoculation methods of plant growth-promoting bacteria.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8826558/
The inoculation method of plant growth-promoting bacteria is an important factor that can affect the colonization of the inoculant in the host plant rhizosphere and impact its downstream effect on plant growth. Seed inoculation is the most widely used on a commercial scale since it is suited to agricultural production and requires less inoculant than the other two under field conditions. To enhance the survival of the bacteria coated on the seeds, a carrier such as peat slurry or a film coat consisting of alginate polymers are often mixed with bacteria during the coating process as a layer protecting inoculants from environmental stresses such as desiccation and temperature perturbations. Soil drench or in-furrow inoculation, on the other hand, is performed by applying the inoculants in soil before or after planting. It has several advantages over seed inoculation as it prevents the inoculants from being inhibited by the chemicals coated on seeds and can be used to deliver inoculants at higher density without being constrained by seed size. Foliar spray and root dipping are two of the most commonly used methods for plant inoculation.
In this report, several inoculation methods ”i.e, seedling priming, soil drench, and three seed coating methods (direct seed coating, alginate seed coating, and 12-h coating)” were compared for their efficacy of delivering three different bacterial strains to sorghum under sterile and field conditions.
Three bacteria isolated from field-grown sorghum were used in this study, with Chitinophaga pinensis (Gram-negative) originating from the root endosphere while Caulobacter rhizosphaerae (Gram-negative) and Terrabacter sp. (Gram-positive) were from the soil.
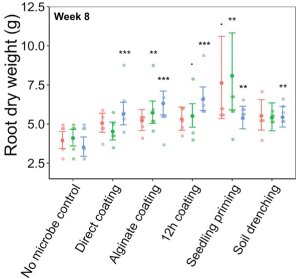
Under Sterile Greenhouse Conditions:
Greater root growth-promotion was detected when inoculating C. rhizosphaerae and C. pinensis with the seedling priming compared to other inoculation methods. In fact, root growth-promotion from C. rhizosphaerae was only detectable with seedling priming despite this effect being marginally significant. For C. pinensis, significant root growth-promotion was also observed with alginate coating and marginally significant for 12 h coating. No significant root growth-promotion was measured when inoculating C. rhizosphaerae and C. pinensis with soil drench method. Among the three bacteria used, only C. pinensis and Terrabacter sp. exhibited significant shoot growth enhancement. Significant shoot growth-promotion from C. pinensis was measured when inoculated with seedling priming, alginate, and 12 h coating methods. Significant shoot growth-promotion was also observed when Terrabacter sp. was inoculated with the same seed coating methods but not the seedling priming.
Under Field Condition:
Two inoculation methods suitable for field planting, alginate and 12 h coating, were tested under field condition. C. rhizosphaerae and C. pinensis were detected in the rhizosphere up to 12 weeks after inoculation in the field, DNA copy numbers in the rhizosphere of the inoculated plants were lower as compared to that of the greenhouse experiment and not significantly different from the uninoculated control, and Terrabacter sp. was not detected in either of the sampling timepoints. This may have been because the bacterial isolate concentration used to coat the seeds (103–104 CFU per seed) was too low to facilitate their establishment in sorghum rhizosphere under non-sterile conditions where there is competition from the natural microbial communities.
Simply coating seeds with a bacterial suspension was suitable for the inoculation and successful colonization of Gram-positive bacteria in the greenhouse, whereas the field results were inconclusive. For Gram-negative bacteria direct inoculation using seedling priming led to higher colonization efficiency than seed coating.