A group from Department of Microbiology, Boston University School of Medicine, Boston, MA, USA, etc. has reported CD169/Siglec-1, a myeloid cell specific I-type lectin, facilitated ACE2-independent SARS-CoV-2 fusion and entry in macrophages.
https://pubmed.ncbi.nlm.nih.gov/35378756/
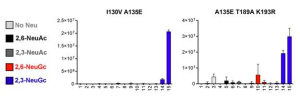
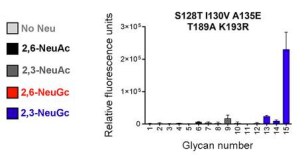
Circulating monocytes, macrophages, and tissue-resident alveolar macrophages are not known to express ACE2, infection receptor of SARS-CoV-2. These cells have been shown to express low levels of TMPRSS2, and moderate levels of endosomal cathepsins which are necessary proteases for SARS-CoV-2 infection. CD169/Siglec-1 (I-type lectin) is highly expressed by splenic red pulp and perifollicular macrophages, subcapsular sinus macrophages and alveolar macrophages. It is also known that CD169/Siglec-1 expression can be upregulated on peripheral blood monocytes under inflammatory conditions, especially in response to type I interferons (IFNs).
In this study, it was examined the mechanisms by which macrophages potentially contribute to inflammation during SARS-CoV-2 infection, using two different human macrophage models, PMA-differentiated THP1 cells (THP1/PMA) and primary monocyte-derived macrophages (MDMs) expressing a myeloid cell-specific receptor, CD169/Siglec-1, in the presence or absence of ACE2.
Pre-treatment with antibodies targeting SARS-CoV-2 S NTD and inhibition of endosomal cathepsins by E64D markedly attenuated S-pseudotyped lentiviral infection in CD169+ macrophages, but a TMPRSS2 inhibitor (Camostat) had no effect, suggesting that SARS-CoV-2 S facilitates endosomal viral entry into ACE2-deficient monocyte-derived macrophages (MDMs).
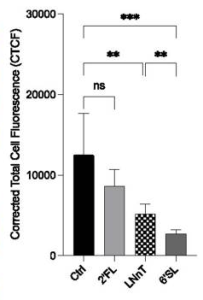
Also, THP1 cells expressing CD169 displayed robust SARS-CoV-2 S binding comparable to levels observed with THP1/ACE2 cells.
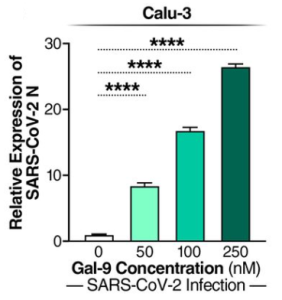
Interestingly, CD169-expressing THP1/PMA cells showed low levels of virus dsRNA production that did not significantly increase over the course of infection. However, significant expression of pro-inflammatory cytokines, IL-6, TNFα, IL-1β, and IL-18, was induced in infected CD169+ THP1/PMA macrophages compared to the parental THP1/PMA cells, indicating that early viral RNA production is the key trigger of innate immune activation, in other words, SARS-CoV-2 infection of macrophages could induce pro-inflammatory responses finally resulting in cytokine storms.