A group from Louvain Institute of Biomolecular Science and Technology, Université catholique de Louvain, Louvain-la-Neuve, Belgium, etc. has reported that multivalent 9-O-Acetylated-sialic acid glycoclusters could be potent inhibitors for SARS-CoV-2 infection.
https://www.nature.com/articles/s41467-022-30313-8
The S1-subunit of SARS-CoV-2 Spike protein can be divided into the N-terminal domain (NTD) containing the glycan-binding domain (GBD) and the C-terminal domain (CTD) accommodating the receptor-binding domain (RBD). The GBD engages glycoproteins and glycolipids in most CoVs8, whereas the RBD binds to the ACE2 receptor.
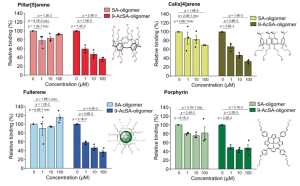
Here authors focused on 9-O-acetylated-SA (9-AcSA), that are of particular interest, since it has been shown that coronaviruses OC43 and HKU bind this glycan via a conserved receptor-binding site in their spike protein.
While monovalent 9-AcSA shows a high IC50 > 100 µM, we observed for all 9-AcSA-derived glycoclusters screened an IC50 in the range 1-10 µM confirming a multivalent effect for these molecules. Among these, 9-AcSA-porphyrin even shows an IC50 in the sub-micromolar range, both on purified 9-AcSA and in the cellular context, making it an excellent candidate as a therapeutic agent.