A group from Kumamoto University, etc. has reported about why Δ mutation (Indian mutant strain named B.1.617) in SARS-CoV-2 has higher infectivity.
https://www.sciencedirect.com/science/article/pii/S1931312821002845?via%3Dihub
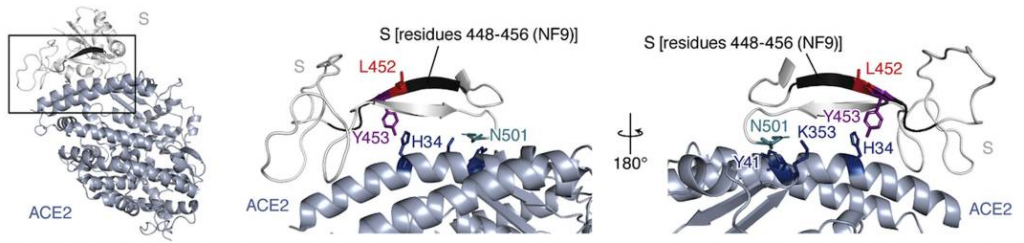
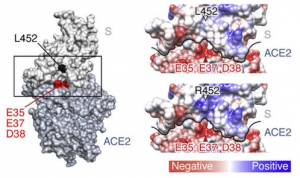
L452R is a key mutation observed in SARS-CoV-2 Δ mutation. It was found that the L452R mutant significantly increased the binding affinity to human ACE2 (Kd = 1.20 ± 0.06 nM). The L452 residue is not directly located at the binding interface (see figure below). Structural analysis and in silico simulation suggested that the L452R substitution promotes electrostatic complementarity.
Because residue 452 is located in close proximity to a negatively charged patch of ACE2 residues (E35, E37, and D38), the electrostatic interaction with ACE2 becomes larger in positively charged R452 than electrically neutral L452 (see figure below).
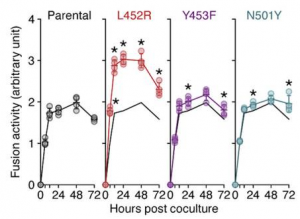
It was also found that the L452R mutation significantly increased fusion efficacy compared to the parental S, using a SARS-CoV-2 S-based fusion assay, suggesting that the L452R mutation promotes viral replication by increasing viral fusogenicity.