A group from Univ. Grenoble Alpes, CNRS, CERMAV, 38000 Grenoble, France, etc. has found a β-trefoil lectin (R-type lectin) in protoctista, Salpingoeca rosetta.
https://www.biorxiv.org/content/10.1101/2022.02.10.479907v1.full
Lectin domains are often associated with other functional proteins such as enzymes or toxins. Life-threatening examples are ricin or cholera toxin, in which the lectin domain is responsible for the specificity and adhesion to cell surface glycans, prior to the cellular uptake of the toxin that interferes with metabolism.
Authors have found a eukaryotic Mytilec domain in the genome of Salpingoeca rosetta for the first time using the TrefLec database, and named it as SaroL-1. It has been kbnown that the Mytilec domain functions as a β-trefoil lectin (R-Type lectin), and binds efficiently to globotriaosyl ceramide (Gb3). β-trefoil lectins (R-type lectins) are widely distributed in bacteria, fungi, plants and animals.
The binding of SaroL-1 to different galactosyl-ligands was assessed in solution by isothermal titration calorimetry. The monosaccharides N-acetylgalactosamine (GalNAc) and α-methyl galactoside (GalαOMe) displayed a similar millimolar affinity with a Kd of 2.2 and 2.8 mM, respectively. All tested αGal disaccharides and the p-nitrophenyl-α-D-galactopyranoside (PNPG) derivative bound with affinities twice as strong, with a Kd close to 1 mM, except for αGal1-4Gal, the terminal disaccharide of the globoside Gb3. Lactose that contains βGal displayed very weak binding, being 20 times less efficient than αGal1-4Gal, confirming the preference for the αGal epitope.
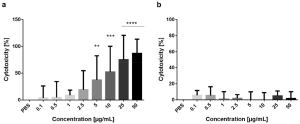
SaroL-1 could form pore-like structures upon membrane binding and work as pore-forming toxins. Actually, SaroL-1 showed strong cytotoxicity towards cancer cells (H1299 cells). The dose-dependent increase of cytotoxicity was confirmed with a standard cell proliferation assay as shown below (a), and cell viability was reduced by approximately 87% after stimulation with SaroL-1. Interestingly, the soluble sugar PNPG could inhibit SaroL-1 cytotoxicity as shown below (b).
This lectin might be a valuable tool for cancer therapy.